 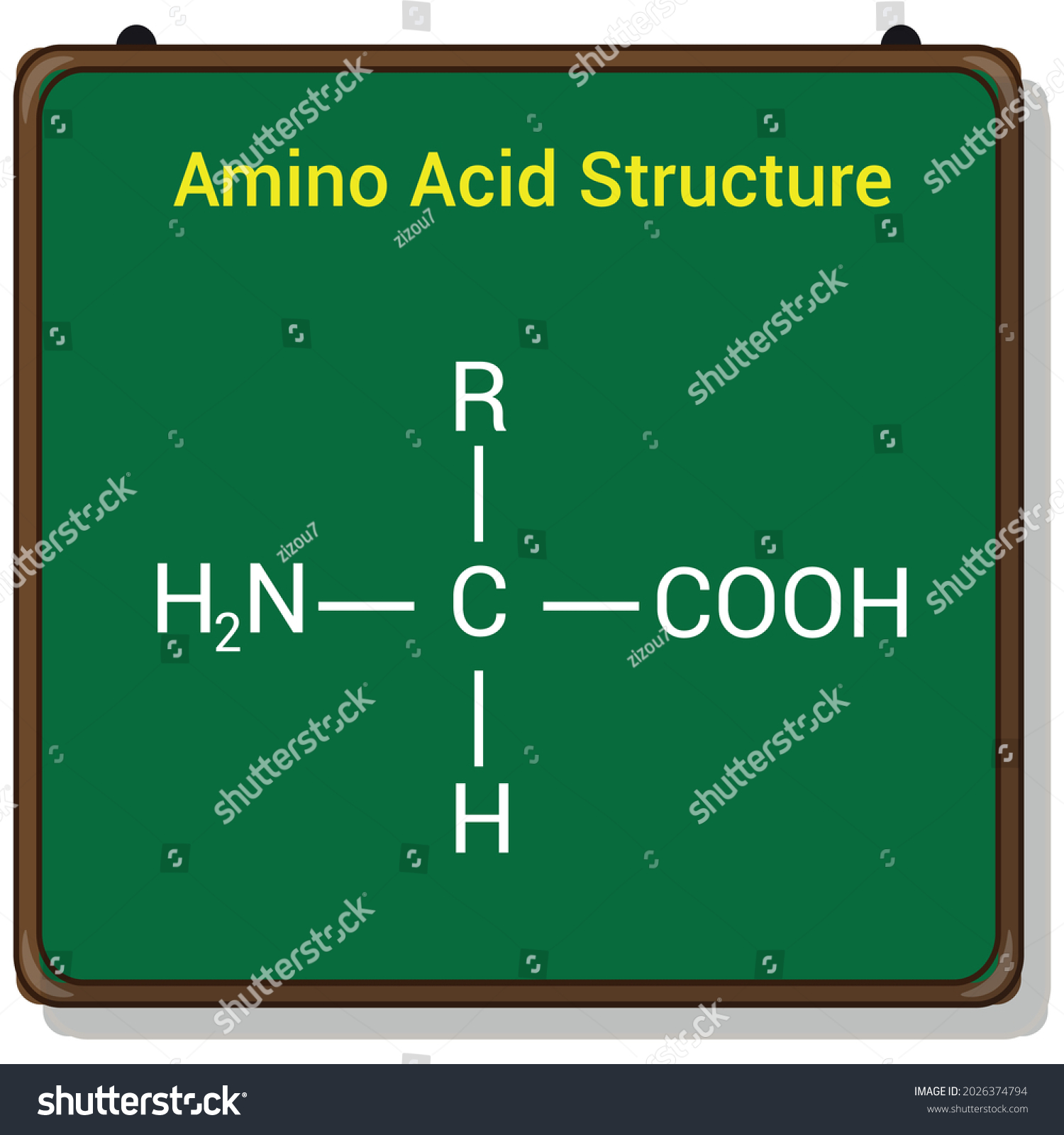 
These charged amino acids play important roles in stabilizing protein 3D structures, and mediating protein-ligand interactions via electrostatic attractions. Two are negatively charged (acidic) and three are positive (basic). Polar and Electrically-charged Amino AcidsĪmong the 20 amino acids, five have a side chain that can be electrically charged at physiological pH. Non-polar amino acids carry a hydrophobic side chain, and are commonly located in the interior of proteins, whereas polar amino acids are typically found on protein surfaces due to the hydrophilic properties of their side chains. The properties of the side chains govern the way they interact with surrounding molecules, and consequently impact the protein’s 3D structure. These amino acids can be broadly grouped based on their biophysical properties ( e.g., charge, hydrophobicity, and polarity of their side chain). Sol: The correct option is “A”. α amino acid is the monomeric units of proteins.DNA encodes twenty types of amino acids that make up proteins in all organisms, from bacteria to plants, to animals. They are generally water soluble and also have high melting points.This point is definite for every α-amino acid. The Isoelectric point is the pH point at which the concentration of zwitter ions is the highest ad the concentration of cationic and anionic form is equal.In aqueous solution, alpha amino acids exist in equilibrium between a cationic form, an anionic form and dipolar ion.This dipolar ion has a special name “Zwitter ions’. Any amino acid in the dry state is in crystalline form.This is the reason they behave like salts. Each amino acid has both an acidic and basic group as you can see from its structure.Now from this information, we can arrive at the properties of amino acids. Now that we have seen the structure and types of amino acids. They are either produced in our bodies or obtained from protein breakdowns. These acids are synthesized in our bodies itself and we need not rely on outside sources for them. We must rely on food sources to obtain these amino acids. These are the acids that cannot be synthesized in our bodies. Some of these neutral acids areĪmino acid can also be classified on the basis of their need to the human body and their availability in the human body 1] Essential Amino Acids Also, they have at least one hydrogen component connected to electronegative atoms. They have an equal number of amino and carboxyl groups. The three basic polar amino acids areĬ) Neutral: These are neither acidic nor basic. These are:ī) Basic: These have an extra nitrogen group that tend to attract a hydrogen atom. These are further divided into three further categories.Ī) Acidic: If the side chain contains an extra element of carboxylic acid component these are acid-polar amino acids. These are also known as Hydrophilic Acids. If the side chains of amino acid contain different polar groups like amines, alcohols or acids they are polar in nature. Numbers one to seven are Alkyl and the last two are aromaticīrowse more about Biomolecules Biomolecule The acids falling in this group are stated below. The R group can be either of Alkyl groups (with an alkyl chain) or Aromatic groups. Now two basic subcategories are 1] Non-Polar Amino Acids This R group makes the amino acid unique.Īmino Acid can be classified based on their structure and the structure of their side chains i.e. This is the variable radical group and is different for every amino acid. The second one is a carboxyl group -OOOH.This atom is surrounded by three chemical groups.All amino acids contain a carbon atom in the middle of the molecule, the alpha-carbon.Although all these have varied structures, the basic structure of amino acid remains uniform. These twenty acids will be our focus here. But only about 20 amino acids form a part of the proteins in the human body. 
There are actually thousands of amino acids occurring in nature. You can download Biomolecules Cheat Sheet PDF by clicking on Download Button Below These are an essential nutrient in our diet because of the functions they perform. This enables the great diversity of proteins that can be found in nature. Amino acid being organic compound molecules can form various different links with each other due to the versatile nature of carbon. These amino acids bond together to form a larger protein molecule. There are some 20 amino acids in the proteins that we consume. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
